Enhancement of Solubility, Purification, and Inclusion Body Refolding of Active Human Mitochondrial Aldehyde Dehydrogenase 2 | ACS Omega

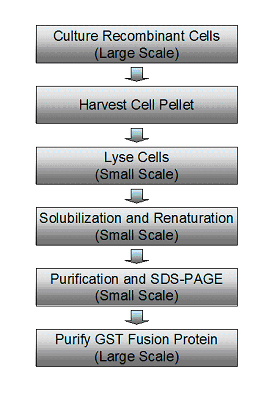
Novel purification strategy for improved recovery of bioactive protein... | Download Scientific Diagram
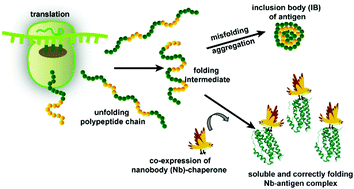
Nanobodies as solubilization chaperones for the expression and purification of inclusion-body prone proteins - Chemical Communications (RSC Publishing)

Inclusion Bodies & Protein Purification Unveiling the Mechanisms for Enhanced Expression & Recovery - YouTube
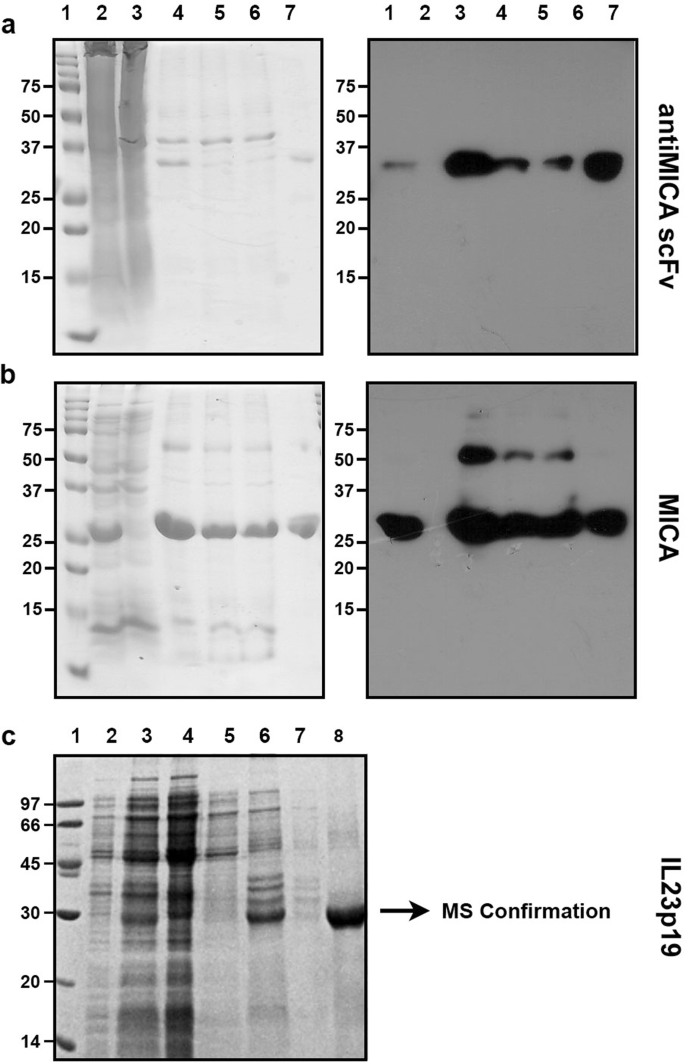
Optimization of culture conditions for the expression of three different insoluble proteins in Escherichia coli | Scientific Reports

Fast Purification of Recombinant Monomeric Amyloid-β from E. coli and Amyloid-β-mCherry Aggregates from Mammalian Cells | ACS Chemical Neuroscience
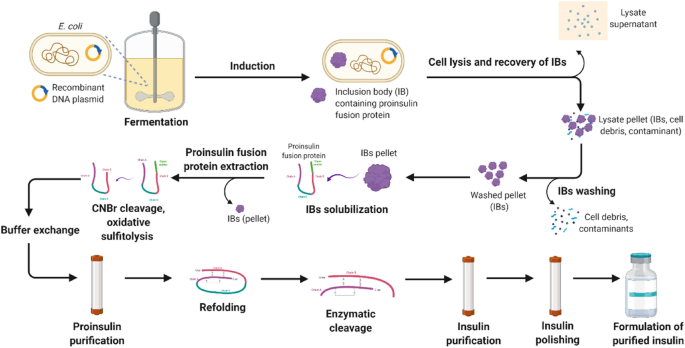
Downstream processing of recombinant human insulin and its analogues production from E. coli inclusion bodies | Bioresources and Bioprocessing | Full Text
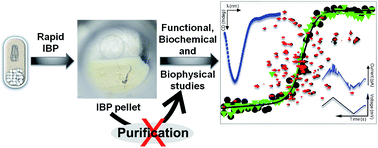
Thermodynamic, structural and functional properties of membrane protein inclusion bodies are analogous to purified counterparts: case study from bacteria and humans - RSC Advances (RSC Publishing)
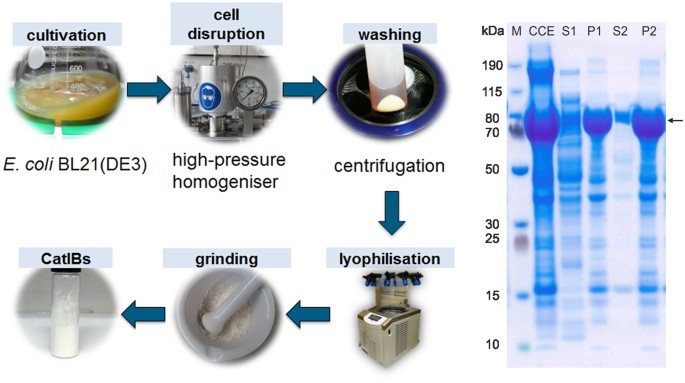
Catalytically active inclusion bodies of L-lysine decarboxylase from E. coli for 1,5-diaminopentane production | Scientific Reports

Wanted: more monitoring and control during inclusion body processing | World Journal of Microbiology and Biotechnology
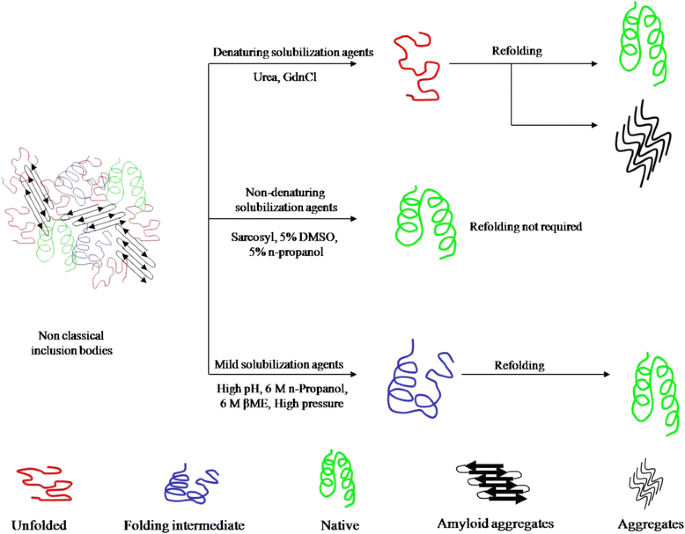
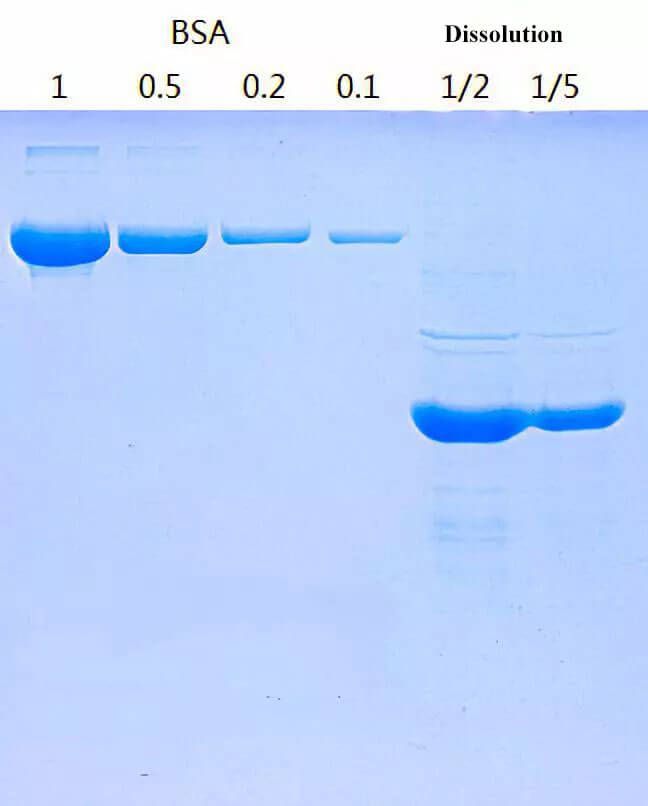
Protein recovery from inclusion bodies of Escherichia coli using mild solubilization process | Microbial Cell Factories | Full Text
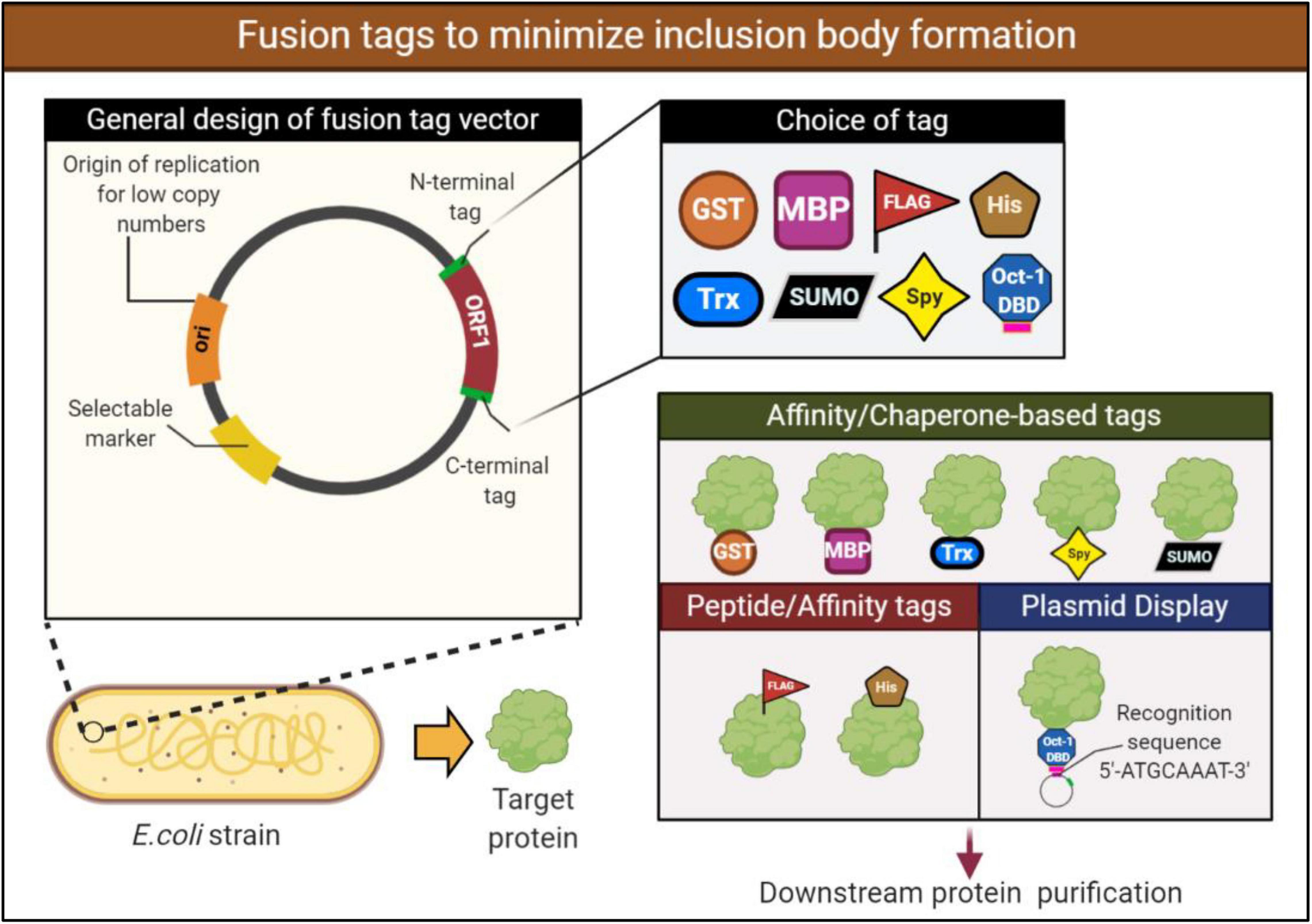
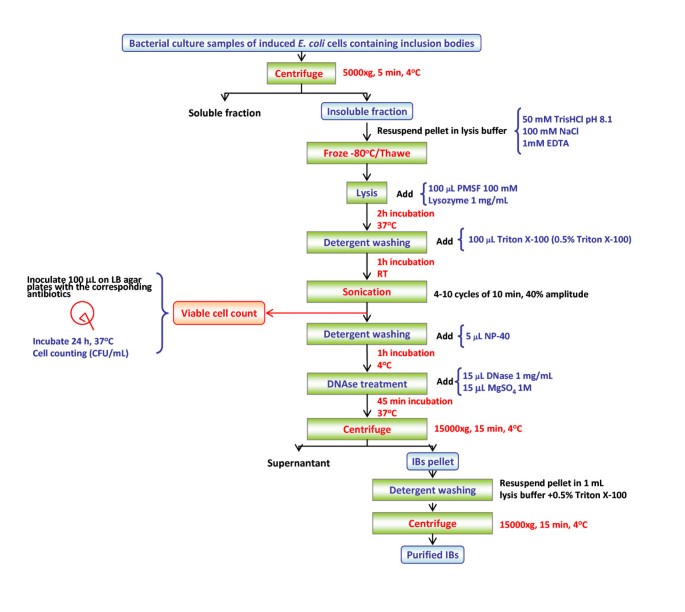
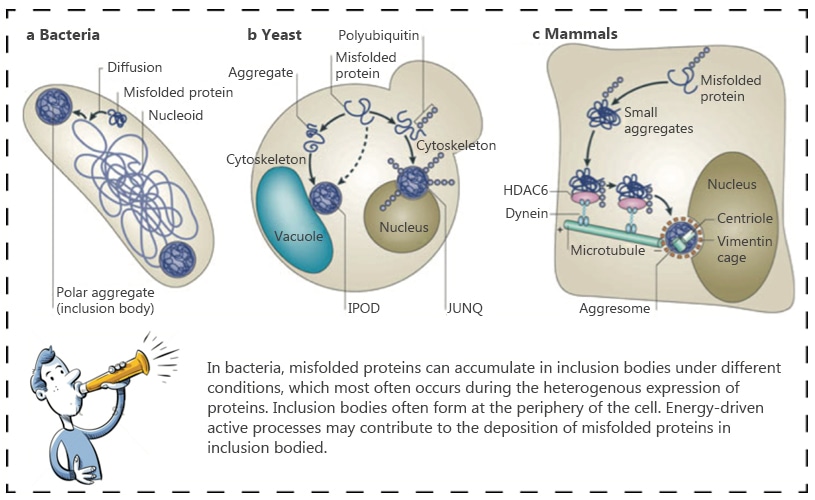
Frontiers | Challenges Associated With the Formation of Recombinant Protein Inclusion Bodies in Escherichia coli and Strategies to Address Them for Industrial Applications
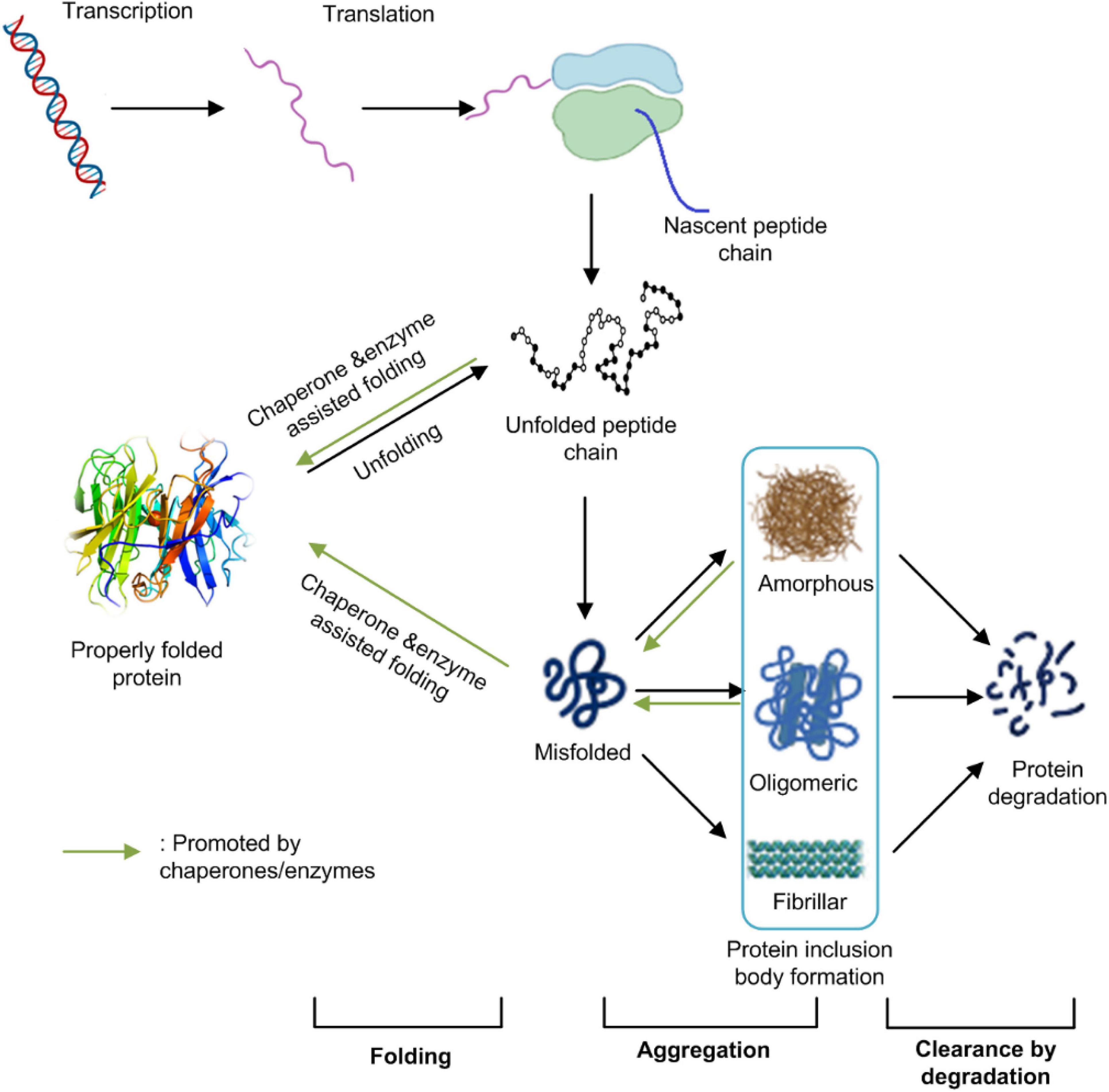
Frontiers | Challenges Associated With the Formation of Recombinant Protein Inclusion Bodies in Escherichia coli and Strategies to Address Them for Industrial Applications

Catalytically-active inclusion bodies for biotechnology—general concepts, optimization, and application | Applied Microbiology and Biotechnology